Decarboxylation
Slovo „dekarboxylace“ může na první pohled nahánět hrůzu, ve skutečnosti je však tento proces, při němž je z organické sloučeniny odstraněna karboxylová kyselina, každodenní realitou pro uživatelé konopí.
Ať už přikládají zapalovač k dýmce napěchované usušenou bylinou, anebo zapínají vaporizér, oběma způsoby zahajují efektivní dekarboxylaci. Tento krok je zásadní v tom, že zpřístupňuje psychoaktivní účinky konopí – bez něj by totiž z kanabinoidních kyselin nevznikaly kanabinoidy, a tudíž v případě THC ani žádný opojný stav.
Jednoduchý chemický proces
Dekarboxylace, tedy reakce rostlinného materiálu konopného původu (v syrovém stavu) se zdrojem tepla, je ve své podstatě velmi jednoduchý chemický proces. Za touto jednoduchostí se však ukrývá složitá a stále se vyvíjející věda, jež v posledních letech pomalu nabývá na důležitosti, což je odrazem faktu, že užívání konopí pro lékařské i rekreační účely se ve společnosti stalo jedním z hlavních témat.
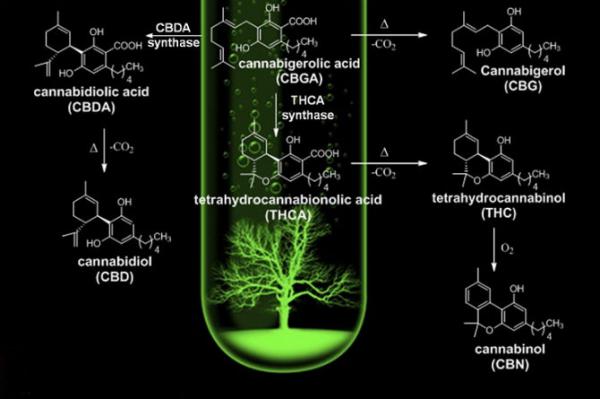
Než spustíme ohněm zapalovače jeho dekarboxylaci, je v konopí přítomna kyselina tetrahydrokanabinolová (THCA) – chemická sloučenina, která vzniká v trichomech na samičích květech této byliny v jejím syrovém stavu a která není ani v nejmenším psychoaktivní. Abychom tedy dosáhli kýženého účinku, je nutné THCA přeměnit na náš starý dobrý tetrahydrokanabinol.
Dekarboxylace probíhá ve dvou základních fázích: buď přirozeně a pomalu, když bylinu sušíme, anebo rychle pomocí zahřívání. Dekarboxylace pomocí kouření či vaporizace nevyžaduje zvláštní řemeslnou zručnost ani soustředění, extrémní žár totiž spustí okamžitou aktivaci THC, které se prostřednictvím kanabinoidních receptorů vstřebává do těla a navozuje omamný stav i léčebné účinky. Pokud však dekarboxylujeme konopí určené například do pokrmů, musíme v něm před samotnou konzumací psychoaktivní kanabinoidy zaktivovat sami – například v horkovzdušné troubě.